
Figure 1.3.3 : The Emission of Light by a Hydrogen Atom in an Excited State. When an atom in an excited state undergoes a transition to the ground state in a process called decay, it loses energy by emitting a photon whose energy corresponds to the difference in energy between the two states (Figure 1.3.1 ). Any arrangement of electrons that is higher in energy than the ground state.: its energy is higher than the energy of the ground state. A hydrogen atom with an electron in an orbit with n > 1 is therefore in an excited state. As n increases, the radius of the orbit increases the electron is farther from the proton, which results in a less stable arrangement with higher potential energy (Figure 2.10). Because a hydrogen atom with its one electron in this orbit has the lowest possible energy, this is the ground state (the most stable arrangement of electrons for an element or a compound), the most stable arrangement for a hydrogen atom. Historically, Bohr’s model of the hydrogen atom is the very first model of atomic structure that correctly explained the radiation spectra of atomic hydrogen. The negative sign in Equation 1.3.3 indicates that the electron-nucleus pair is more tightly bound when they are near each other than when they are far apart. The orbit with n = 1 is the lowest lying and most tightly bound. In his final years, he devoted himself to the peaceful application of atomic physics and to resolving political problems arising from the development of atomic weapons.Īs n decreases, the energy holding the electron and the nucleus together becomes increasingly negative, the radius of the orbit shrinks and more energy is needed to ionize the atom. (b) The energy of the orbit becomes increasingly less negative with increasing n.ĭuring the Nazi occupation of Denmark in World War II, Bohr escaped to the United States, where he became associated with the Atomic Energy Project. The Bohr model, named after scientist Neils Bohr, is a way of illustrating the structure of the atom and the location of its subatomic particles. Figure 1.3.2: The Bohr Model of the Hydrogen Atom (a) The distance of the orbit from the nucleus increases with increasing n. In this state the radius of the orbit is also infinite. Bohr was also a philosopher and a promoter of scientific research. In that level, the electron is unbound from the nucleus and the atom has been separated into a negatively charged (the electron) and a positively charged (the nucleus) ion. Niels Henrik David Bohr (Danish: nels po 7 October 1885 18 November 1962) was a Danish physicist who made foundational contributions to understanding atomic structure and quantum theory, for which he received the Nobel Prize in Physics in 1922. 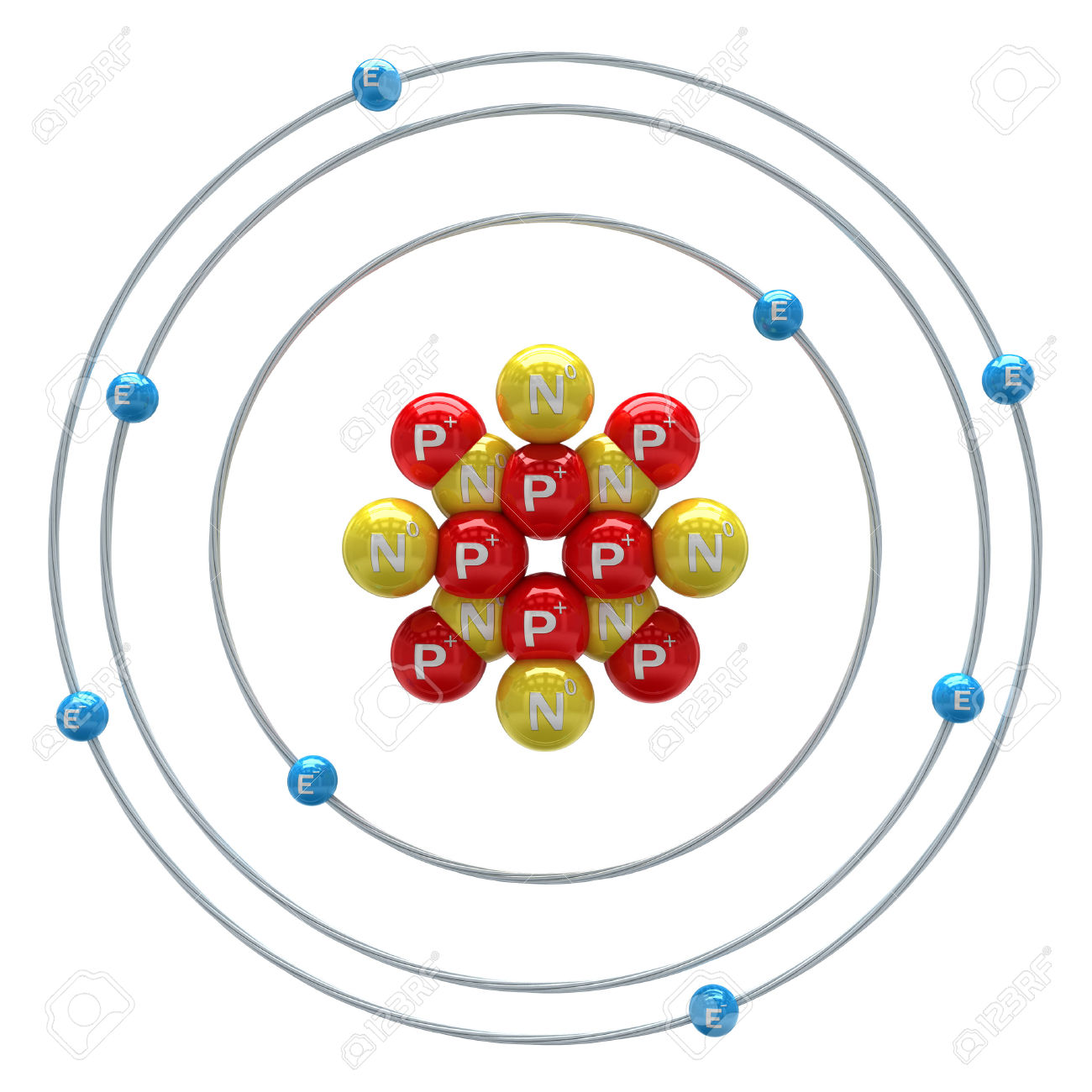
Metal coat hangers make good orbitals and sheets of aluminum foil make a good wave model of an atom. Styrofoam balls are a popular choice, but you can use cotton balls, marbles, small balls of aluminum foil, beads or even candy. In this model n = ∞ corresponds to the level where the energy holding the electron and the nucleus together is zero. You need balls for the nucleus, and possibly, for the electrons. \(n\) is a positive integer corresponding to the number assigned to the orbit, with n = 1 corresponding to the orbit closest to the nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
